ISO 62366-1 -standard
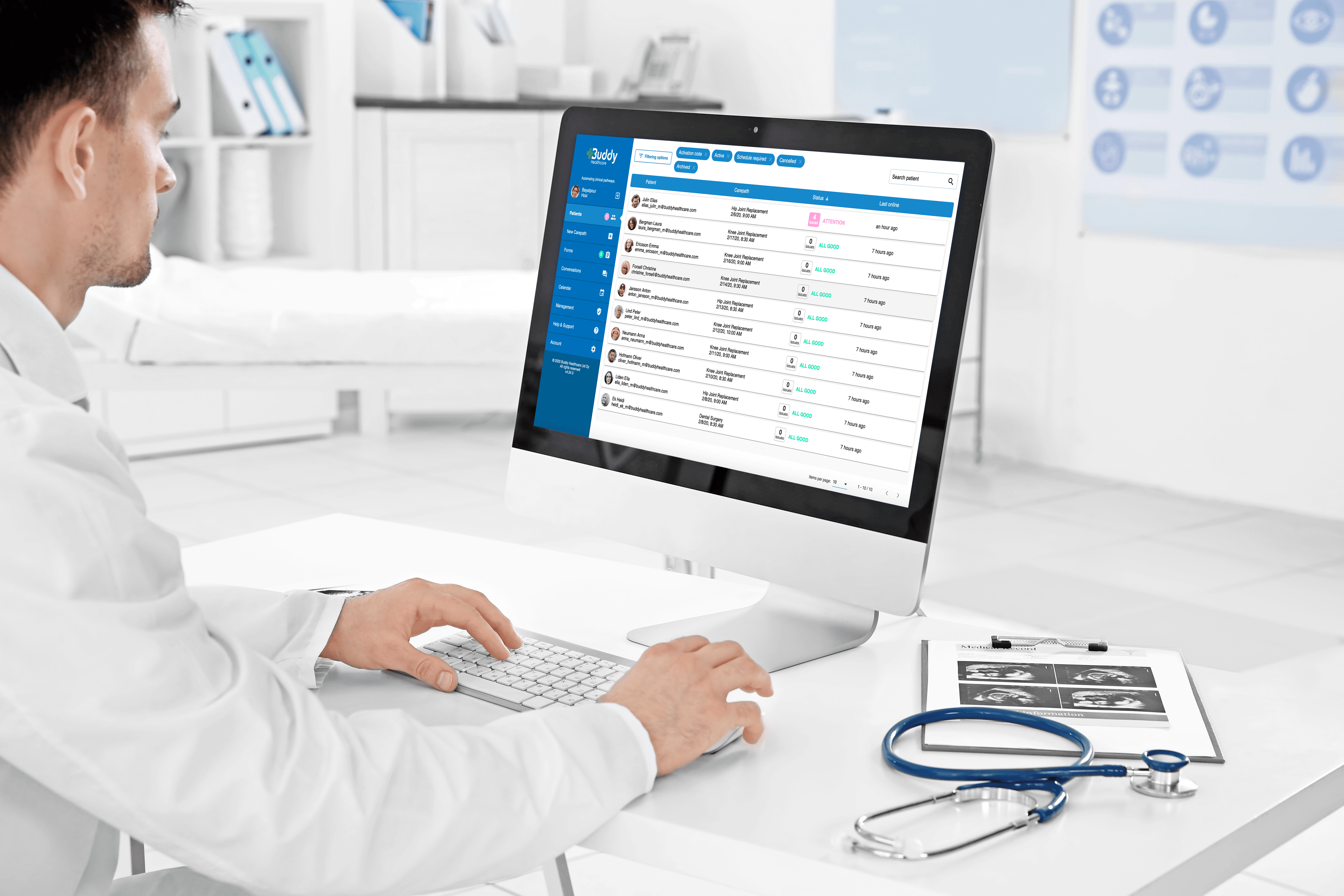
IEC 62366-1:2015 describes the processes for a manufacturer of medical devices to analyse, specify, develop and evaluate the usability of the medical device as it relates to safety.
What is IEC 62366-1?
IEC 62366-1:2015 describes the processes for a manufacturer of medical devices to analyse, specify, develop and evaluate the usability of the medical device as it relates to safety. The usability engineering process i.a. Human factors engineering guides the manufacturer to identify, assess and mitigate usability risks associated with normal use i.a. correct use and possible use errors and gives the tools to control these risks. The standard is best used together with EC/TR 62366-2 since it provides background information on the methods and hands-on guidance on the implementation of the standard.
Together with Kasve you ensure that the usability of your medical device will be addressed in appropriate level in all of the product development phases and that the requirements of the standard will be fulfilled.
Looking for a quality expert?
We have helped a number of medicinal device manufacturers, testing and clinical laboratories, and start-ups develop their operations.